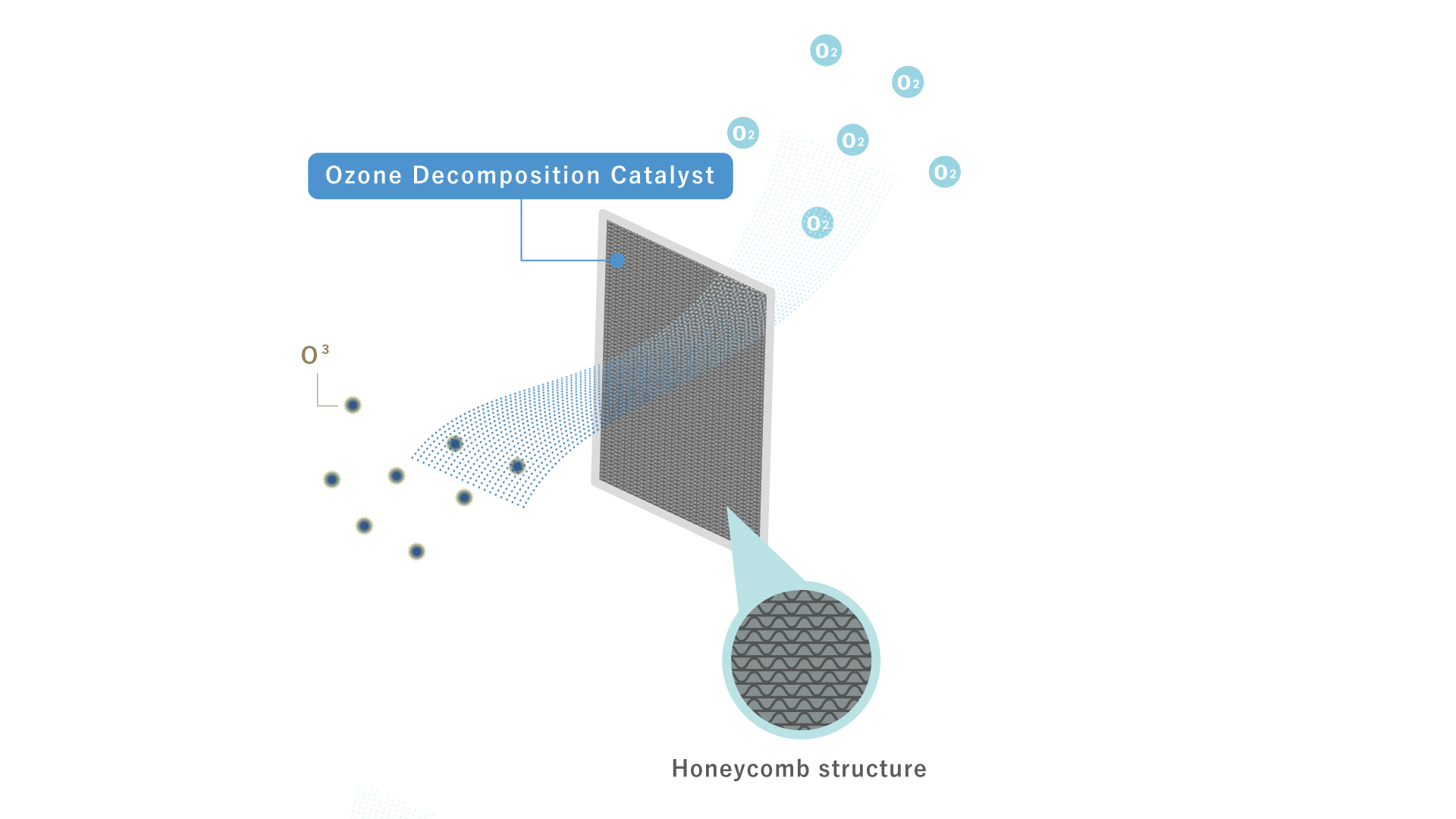
Ozone Decomposition Catalyst

| Model | NHC-M (metal honeycomb type)
NHC-R (ceramic honeycomb type) |
|---|
Decomposes harmful ozone into harmless oxygen! A high-performance catalyst for long-term use.
Product Overview
This catalyst uses a honeycomb structure. At room temperature or lower, ozone, which is harmful to the human body, is efficiently decomposed into oxygen and rendered harmless.
Features
- Long-term continuous use
- Catalyst specifications can be chosen according to intended purpose
- Decomposes and processes ozone at a high speed
Specifications
Select the size, cell density, and more to suit the catalyst's intended use.
| Type | NHC-M series | NHC-R series |
|---|---|---|
| Material | Metal | Ceramic |
| 特徴 |
|
|
| Size |
Standard sizes:
*Also available in compatible sizing |
Use
Ozone decomposition filters for copy machines, ozone treatment for electrostatic precipitator exhaust, and more.
What is ozone? Does it really affect the human body?

When people think of ozone, most think of the ozone layer. About 90% of the ozone in the atmosphere stays in the stratosphere (about 10~50 kilometers above the ground), absorbing harmful ultraviolet rays from the sun and protecting ecosystems on Earth. Ozone is also produced in the troposphere, which is about 15 kilometers above the ground, but contrary to its role in the stratosphere, this ozone is harmful to the human body. Ozone is a gas composed of three oxygen atoms (O3), and it is characterized by its strong oxidizing properties and its ability to react with many substances. This trait is used for disinfection, sterilization, deodorization, and water treatment, but when ozone over a certain concentration enters the body, all of the mucous membranes it passes through, including the nasal passages, throat, trachea, and lungs, are oxidized. This can result in odor, irritation, coughing, headaches, drowsiness, chest tightness, and other bodily harm. Continued inhalation of high concentrations of ozone can reach deep into the lungs and cause respiratory problems such as pulmonary edema. As such, the Japan Sociey for Occupational Health and the Japan Industrial Safety and Health Association have established an inhibitory concentration of 0.1ppm as a working environment standard.
| Ozone concentration | Effects on living organisms |
|---|---|
| 0.01~0.02 ppm | Noticable odor |
| 0.1 ppm | Strong odor, irritation to the nose and throat |
| 0.2~0.5 ppm | In 3~6 hours, loss of vision will occur |
| 0.5 ppm | The upper airway feels distinctly irritated |
| 1~2 ppm | In 2 hours, headache, chest pain, upper airway dryness, and coughing will occur |
| 5~10 ppm | Increased heart rate and pulmonary edema |
| 15~20 ppm | Within 2 hours, small animals will die |
| 50 ppm | Within 1 hour, a human's life is in danger |
How can ozone be removed?
Catalyst methods are effective in removing ozone from the atmosphere to prevent the aforementioned health hazards. This catalyst is effective at temperatures below room temperature. When ozone comes into contact with the catalyst's surface, it can be detoxified by decomposition into oxygen. The conflicting issues of catalytic efficiency and pressure loss are solved by adopting a honeycomb structure, resulting in high performance and low loss of pressure. This allows for superior performance over long periods of time, even in environments with relatively high wind velocities, such as office equipment. VOC countermeasure specifications with environmental regulations for office equipment are also available separately.
Ozone decomposition reaction mechanism:
Step1:O₃ + M(Catalyst) → M-O + O₂
Step2: M-O + O₃ → M + 2O₂
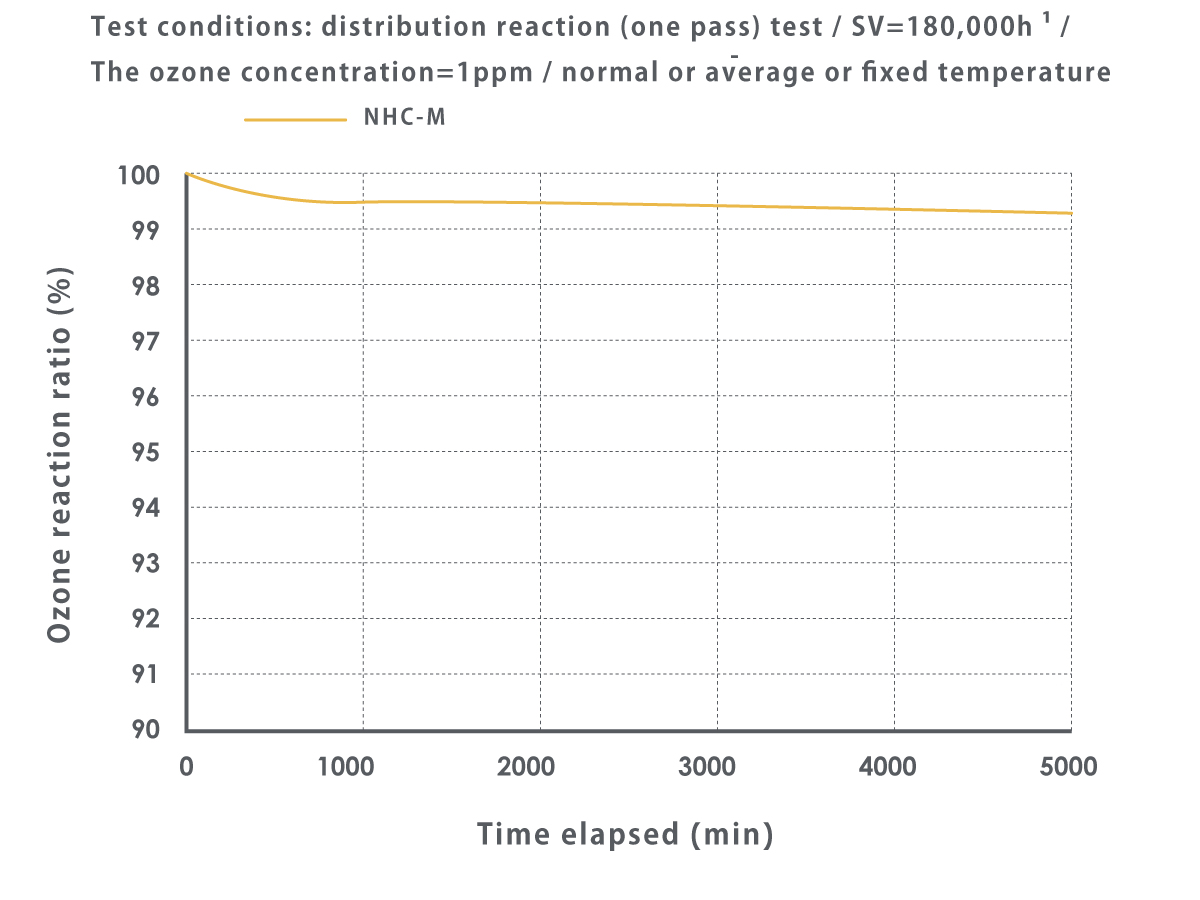
How does the catalyst change the ozone concentration?
Contact Sales Department
| Department | Business Innovation Office |
|---|---|
| Contact Form | |
| TEL | 81-3-5436-8484 |
| FAX | 81-3-5436-8680 |

